Electron charge can be abbreviated as e for example, 1 electron charge can be written as 1 e. It is equal to 1.602176634×1019 coulombs, per the 2019 SI redefinition of the coulomb. To help show this three-dimensional shape even more accurately, we can rely on space-filling models as well as ball-and-stick models. Electron charge is equal to the charge of an electron, and is the inverse of elementary charge, which is the magnitude of the charge of a proton. We will discuss the significance of these electrons at the end of this section. The two dots above nitrogen indicate a lone pair of electrons that are not involved in any covalent bond. 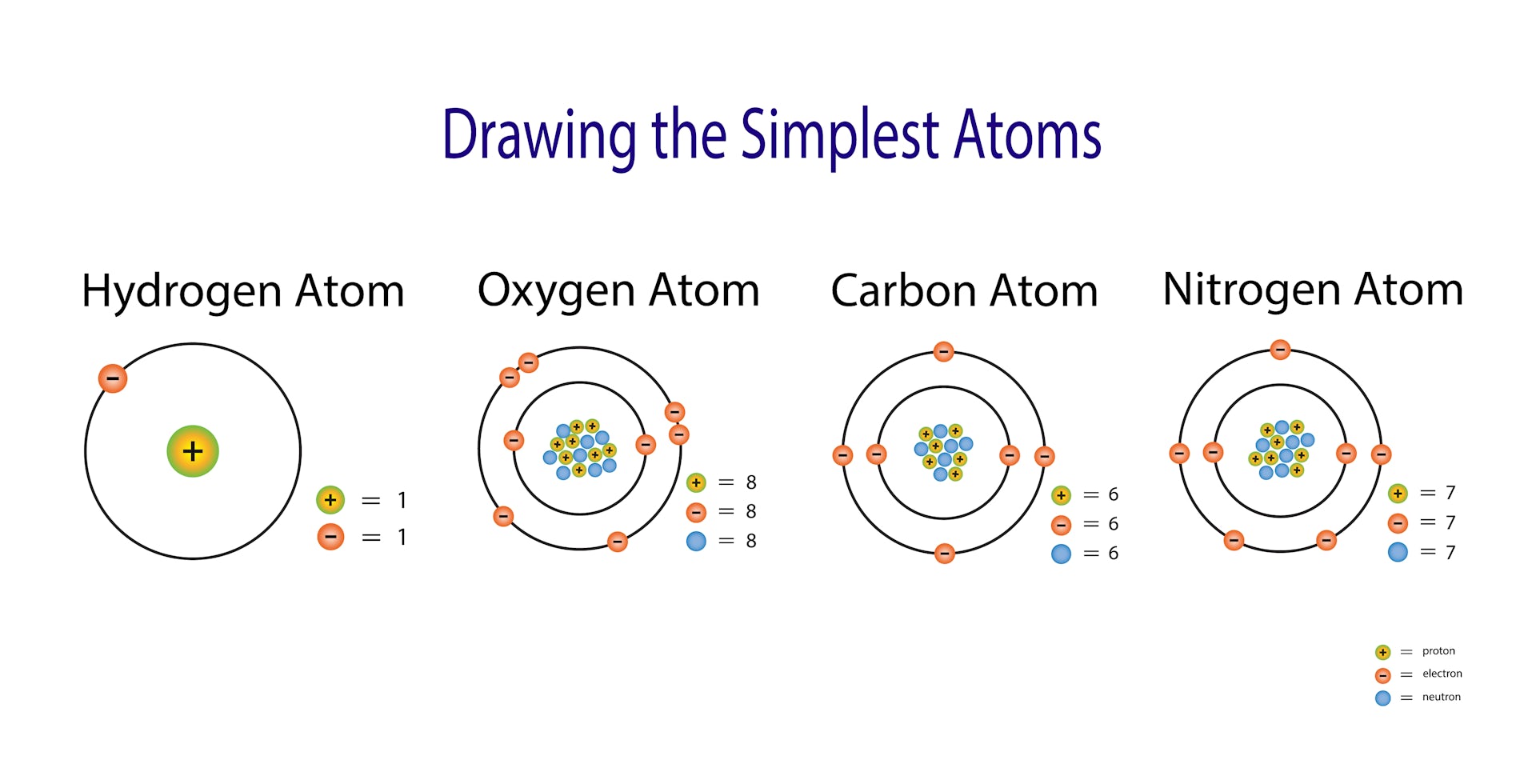
what is the total charge on 1 mole of electronif there is same amountof charge on one proton, calculate total charge on 1 mole of protons. charge of one electron is 1.6022 10-19 coulomb. However, in the more detailed structural formula on the right, we have a dashed line to indicate that the rightmost hydrogen atom is sitting behind the plane of the screen, while the bold wedge indicates that the center hydrogen is sitting out in front of the plane of the screen. Charge of one electron 1.602 × 10 19 coulomb Q. In the structural formula to the left, we are only seeing a two-dimensional approximation of this molecule. Keep in mind, however, that atoms and molecules, just like everything else in the universe, exist in three dimensions-they have length and width, as well as depth. As we know, the Charge on one electron is equal to. One mole of electrons is required for the reduction of one mole of ions. Mass of neutrons in 18 mL of water = 8.0695 x 10 -3 kgFrom both of these structural formulas, we can see that the central nitrogen atom is connected to each hydrogen atom by a single covalent bond. When supposing y moles of electrons are passed through three cells, the mass of sodium, aluminium and copper liberated are 23y grams, 9y grams, 31.75y grams respectively. Mass of neutrons in 18 mL of water = 1.675 x 10 -27 x 4.8176 x 10 24 Using the faraday constant, we can then change the charge (C) to number of moles of electrons transferred, since 1 mol e- 96,500 C. Number of neutrons in 1 molecule of water = 8 + 0 = 8 Number of neutrons in 2 hydrogen atoms = 0 x 2 = 0 Number of neutrons in 1 atom of hydrogen = 1 – 1 = 0 Mass of water = Volume x density = 18 x 10 -3 L x 1 kg/L = 18 x 10 -3 kg = 18 g Given volume of water = 18 mL = 18 x 10 -3 L Radius of carbon atom = (1.2 x 10 -8)/2 = 6 x 10 -9 cm = 6 x 10 -11 m = 0.6 x 10 -10 m = 0.6 angstromįind the total number and total mass of neutrons in 18 mL of water. Number of electrons in 1 gram = 1/(9.1 x 10 -28) = 1.098 x 10 27 electronsĢ x 10 8 atoms of carbon are arranged side by side, calculate the radius of carbon atom, if the length of this arrangement is 2.4 cmĭiameter of carbon atom = 2.4/(2 x 10 8) = 1.2 x 10 -8 cm Hence there is no effect o change of temperature and pressure.Ĭalculate the number of electrons which will together weigh 1 gram. Mass of protons in 34 mg of ammonia = 2.015 x 10 -5 kgĭue to change in temperature and pressure, there is no change in number of moles of the gas. Number of molecules in 34 mg of ammonia = 2 x 10 -3 x 6.022 x 10 23 = 1.2044 x 10 21 Q I.t where Q charge (C) I current (A) t time (s) Q n.F where Q charge (C) n number of moles of electrons (mol) F Faradays constant (96500 C. Number of protons in 1 molecule of ammonia = 7 + 3 = 10 Number of protons in 3 hydrogen atoms = 1 x 3 = 3 Number of protons in 1 atom of hydrogen = Atomic number = 1
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
